Category: Regulations
-

Prescription Insomnia App Okayed by FDA
A mobile app prescribed by physicians to treat chronic insomnia is cleared by the Food and Drug Administration for marketing in the U.S.
-
Trial to Test Arthritis Drug for Covid-19 Pneumonia
A clinical trial is beginning soon that tests a current drug for rheumatoid arthritis as a treatment for severe pneumonia in patients with Covid-19 infections.
-

FDA Okays Faster Covid-19 Test, Nixes Home Tests
The Food and Drug Administration granted an emergency clearance for a point-of-care test to detect for the virus causing novel coronavirus or Covid-19 infections.
-

FDA Outlines Trial Disruption Steps Due to Covid-19
The Food and Drug Administration outlined steps clinical trial teams should take if their studies face disruptions from the novel coronavirus, or Covid-19, pandemic.
-

FDA Clears High-Volume Covid-19 Test
The Food and Drug Administration cleared for use in the U.S. a high-volume diagnostic for the novel coronavirus that can run on automated systems already in place.
-
FDA Okays MRI with Epilepsy Brain Stimulation
A company making brain stimulation systems to treat epilepsy received regulatory clearance for MRI scans in people with its implanted devices.
-

Skin Cancer Apps Shown Misleading, Unreliable
A review of smartphone apps that photograph and purport to analyze skin conditions for signs of cancer shows the apps are unreliable in detecting cancer.
-
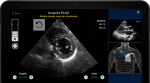
A.I. Heart Ultrasound Software Cleared by FDA
Software guided by algorithms that capture top-quality ultrasound images of the heart is now authorized by the Food and Drug Administration.
-

FDA Approves Children’s Peanut Allergy Drug
The U.S. Food and Drug Administration on Friday approved the first treatment for children to reduce severe allergic reactions to peanuts.
-

FDA Clears A.I. Algorithms for Stethoscopes
Algorithms used in analytical software for digital stethoscopes to diagnose irregular heart rhythms received marketing clearance from the U.S. Food and Drug Administration.