Category: Regulations
-
FDA Blocks Monthly HIV Drug Combo
The U.S. Food and Drug Administration rejected a combination of experimental and existing drugs given once a month to treat HIV.
-
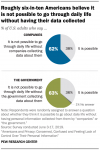
Infographic – US Public Says It’s Tracked Each Day
A solid majority of Americans says companies or government agencies are collecting data about them each day, and they’re not happy about it.
-
Patient Confidentiality, An Issue for New Medical Businesses
How are you going to keep your patient files confidential, and still ensure you’re running with all the access and affordability you need to?
-

Bioprinting Ear Tissue Tagged Rare Disease Treatment
A company developing 3-D printed outer ear tissue received a rare pediatric disease designation on its treatment designed for children.
-

Anti-Vaccine Groups More Adept at Facebook Ads
A review of advertisements about vaccinations shows a few groups took advantage of Facebook’s advertising policies to spread vaccine misinformation.
-

Safety Reports Analyzed with A.I. to Reduce Medical Errors
Research is underway to write algorithms for analyzing free text and other unstructured data in safety report databases to reduce medical errors.
-

Opioids Provide Few Pain Benefits, Yet Abuse Growing
Opioids appear to provide little benefit for patients with pain from osteoarthritis, yet opioid use disorder rates among patients with painful conditions continue to rise.
-

Infographic – U.S. Rated Best in Pandemic Prep
A new report on global health security says no country in the world is fully prepared to handle pandemics or epidemics, with data from the report displayed in this weekend’s infographic.
-
FDA – New Processes, Incentives Needed for Drug Shortages
A report from the Food and Drug Administration identifies the causes of recent shortages of some drugs, and calls for new processes and incentives to prevent future shortages.
-
FDA Tags Biofilm Disruption Device as Breakthrough
A system using an electric current to break up bacterial communities on orthopedic implants received a breakthrough-device designation from the Food and Drug Administration.