Category: Regulations
-
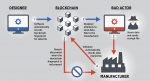
Blockchain Manufacturing Model Described
A report from the U.S. government’s standards agency offers a model for using blockchain, a distributed transaction process, to secure transmissions in smart manufacturing systems.
-

Insulin Delivered by Capsule, That’s the Good News
Before we start celebrating the end of injections, we need to deal with a more basic problem facing people with diabetes, namely the rapidly rising cost of insulin.
-

Trial Testing Vaginal Ring, PrEP in Teens, Young Women
A clinical trial is underway testing use and preferences for drugs or a medical device to prevent HIV infection among young women and teenage girls in Africa.
-

Book Review – The Fifth Risk
Michael Lewis describes in a gripping narrative some of those critical life-protecting functions the federal government performs in his new book The Fifth Risk.
-

Shutdown Delays FDA Peanut Allergy Drug Review
Regulatory review of a drug to prevent allergic reactions to peanuts appears to be delayed by the shutdown of some U.S. government operations.
-

AI, Wearables Posing Health Privacy Risks
An analysis shows artificial intelligence algorithms can connect Americans’ activity tracking data to to their individual private medical information.
-

Duchenne Stem Cell Trial Halted
A clinical trial testing stem cell treatments for symptoms of Duchenne muscular dystrophy is on hold after a patient reported a severe reaction.
-

Govt. Shutdown Disrupts U.S. Science
Several agencies that run or finance labs are closed, due to the lapse of funding for about a quarter of federal departments that began on 22 December.
-
FDA Clears Inhaled Parkinson’s Drug
A drug administered as an inhaled powder to supplement a common treatment for Parkinson’s disease received approval for marketing from the Food and Drug Administration.
-

CDC Finds 12 People Sickened by Stem Cell Provider
Centers for Disease Control and Prevention issued a report on a provider of stem cell therapies indicating 12 people in 3 states were infected with bacteria from that company’s treatments.