Category: Regulations
-
Trial Testing Nerve Stimulation for Stroke Rehab
A clinical trial is recruiting participants to test an implanted device that electronically stimulates a key nerve pathway for restoring use of a person’s arm after suffering a stroke.
-
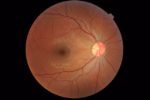
FDA Clears A.I. Software Detecting Diabetic Eye Disease
The Food and Drug Administration authorized a system for detecting diabetic retinopathy, a condition threatening the vision of people with diabetes, that uses artificial intelligence to analyze images of patients’ retinas.
-
Complications Can Result from Plastic Surgery Tourism
Surgeons at a Boston hospital report a continuing flow of cases where people go overseas for plastic surgery, only to encounter complications needing repair, including drug-resistant bacterial infections, after they return.
-
FDA Authorizes Interoperable Glucose Monitoring System
The Food and Drug Administration authorized a device that electronically monitors blood glucose levels in people with diabetes, but is also designed to exchange data with other compatible electronic systems.
-
High False-Positive Rate Found in Consumer Genetic Tests
Follow-up reviews of results from direct-to-consumer genetic tests show 40 percent of the actionable gene variations reported in the tests, including some indicating disease risk, were erroneous.
-
Trials Show Pain Drug Reduces Post-Surgery Opioid Use
Initial results from two late-stage clinical trials show an experimental pain drug with non-opioid ingredients reduces the use of opioid pain relievers following surgery, compared to a placebo and standard care.
-
Quick Bacterial Blood Detection Gains Breakthrough Tag
A technology that detects dangerous bacteria in patients’ blood samples in less than an hour received a breakthrough medical device designation from the U.S. Food and Drug Administration.
-
Trial Shows Patch Feasible for Heart Rhythm Detection
Results from a clinical trial show a wearable patch, applied and used by older adults in their homes, detects more heart rhythm problems than waiting to see a doctor, the usual way these problems are detected.
-
FDA Clears Consumer Genetic Breast Cancer Risk Tests
The Food and Drug Administration authorized the first direct-to-consumer tests of genetic risk for cancer, in this case 3 variations of genes associated with higher risk of breast and ovarian cancer.
-
Trial Shows Reduction in Huntington Disease Protein
First results from a clinical trial show an experimental therapy reduces production of a protein damaging nerve cells in people with Huntington disease.